materials and methods |
materials and methods
|
|
|
Previous studies have demonstrated the MDSC differentiation is possible in vitro, although morphologically correct granulocytic MDSCs have not been produced. In order to both confirm that MDSCs can be differentiated in vitro and determine if those differentiated cells are functionally and morphologically similar to MDSCs produced naturally in the body, BALB/c bone marrow-derived stem cells were grown in BALB/c 4T1 medium and a qualitative analysis of flow cytometry was performed.
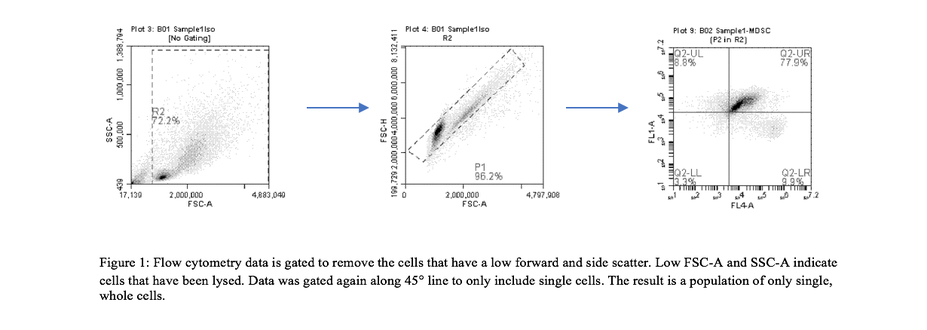
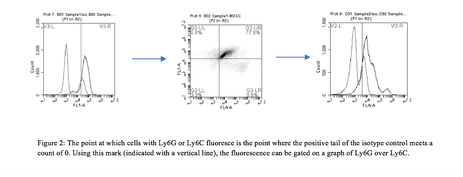
4T1 conditioned media was synthesized. In order to obtain the correct ratio of cytokines required for MDSC differentiation, 4T1 cells were grown for four weeks with RCMI 1640 cell culture medium. RPMI 1640 medium used contained 10% FBS, 1% sodium pyruvate, 1% HEPES, 1% penicillin streptomycin, and 86% basil RPMI. After incubating cells at 37°C, sufficient cytokines necessary for differentiation were assumed to be produced. MDSC differentiation. Bone-marrow derived stem cells were extracted from euthanized BALB/c mice femurs and tibias.[1] The immature cells were treated with ACK lysing buffer to selectively remove red blood cells and left in culture for three days until remaining B and T cells died. The remaining growth media was aspirated and 12mL of growth media containing 60% RMPI 1640 and 40% 4T1 conditioned medium was added. The MDSC precursors were left to grow at 37°C for seven days. Antibody probing/Flow cytometry preparation. In order to probe the differentiated MDSCs, we followed the Pullen Lab protocol for MDSC antibody conditioning. MDSCs were obtained by scraping the plate where the phenotypically attached cells were growing. About 5x105 cells were needed. The cells were then centrifuged at 500g for 5 minutes. The medium was aspirated until the only the pellet of cells remained. The pellet was then resuspended in 800ml of incubation buffer. The cells were centrifuged again as mentioned previously. Cells were aspirated, resuspended, and centrifuged as before. CD11b, Ly6C, and Ly6G receptors were blocked by adding blocking antibody solution to the pellet. Cells were incubated on ice for five minutes. 700ml of incubation buffer was added and the suspension was centrifuged for five minutes. The supernatant was aspirated and PE-conjugated antibodies were added to their respective plates (antibody solution or isotype control). Because the antibodies are light reactive, cells were incubated on ice in the dark for one hour. 700ml of incubation buffer was added, cells were centrifuged, and aspirated. Cells were once again washed with incubation buffer and centrifuged. After aspirating media, the cells were resuspended with 500ml of incubation buffer. 10ml of cells were obtained and injected onto a Conuntess slide to determine the amount of remaining cells. Flow cytometry and analysis of data. Flow cytometry is a technique used to count individual cell types and analyze their size and complexity by hydrodynamically focusing a cell suspension to obtain a single cell stream. Flow cytometry was performed on both the isotype and experimental groups. In order to analyze the data, flow side scatter (SSC-A) over forward scatter (FSC-A) data were gated to only include non-lysed, single cells (figure 1). Cells with CD11b were gated. Isotypes for Ly6G and Ly6C were compared to experimental groups to determine the point at which MDSCs fluoresce. Using that information, data was graphed according to Ly6G and Ly6C fluorescence to determine the amount of G-MDSCs and M-MDSCs (figure 2).
|
Acquisition of pluripotent stem cells from healthy black-6 mouse. Black-6 stem cells were obtained from an individual without tumor. The tibia and femur from each leg were dislocated from the animal and the surrounding muscle was removed. A 25-gauge needle and syringe with suspension media was inserted into the marrow cavity of each bone, and after the syringe was emptied, bone marrow was separated from the leg bones. ACK lysing buffer was added to the cells to selectively destroy red blood cells and reticulocytes. Only bone marrow and adult lymphocyte (B and T cells) precursors will remain, however lymphocytes will die within a few days outside of the body. Cells were counted using the Countess. They were then grown in growth medium (60% RPMI 1640 and 40% 4T1 conditioned medium) and incubated at 37°C for seven days.
Differentiation of black-6 MDSCs with BALB/c conditioned media. Antibody probing and flow cytometry data collection and analysis were performed using the same methodology as described in the previous section. evaluation of IL-6 cytokineBecause of the inability to access large quantities of MDSCs from cancer patients, not much is known about MDSCs or the cytokines required to differentiate them. An enzyme linked immunosorbent assay (ELISA) could be used to detect the presence of IL-6 secreted from 4T1 which may indicate that IL-6 is necessary for differentiation. The presence of IL-6 in 4T1 medium led to a concentration assay using varying amounts of IL-6 neutralizing assay and IL-6 peptide.
IL-6 ELISA. A capture antibody comprised of 21 µl Rabit Anti-Muring IL-6 + .5mg D-mannitol centrifuged and reconstituted in 210 µl sterile water was diluted with 100 µl 1xPBS and 100 µl of this solution was added to each ELISA plate well. The blocking buffer solution was incubated overnight at room temperature. The wells were aspirated, washed with wash buffer (0.05% Tween-20 in PBS), and dried. 300 µl of block buffer comprised of 1% BSA in PBS was added to each well. The plate was incubated for one hour at room temperature. The standard row of wells was also prepared. A standard was diluted from 4000pg/ml to 0 using diluent made of 0.05% Tween-20, 0.1% BSA in PBS. 100 µl of each standard was added to each well in triplicate. In order to detect the IL-6 present, a diluted detection antibody (3µl ig Biotinylacted Rabbit Anti-Murine IL-6 + 2.2mg D-mannitol reconstituted in 30µl sterile water was diluted to 0.1µl/ml. 100µl detection antibody was added to every well. The plate was incubated at room temperature in the dark for thirty-five minutes for color development. 100 µl of IM HCl Stop Solution was added. The color development was analyzed with an ELISA plate reader at 450nm with a wavelength correction set at 620nm. IL-6 depletion assay. 33mL of growth medium comprised of 37.6% RPMI-1640, 10% Fetal Bovine Serum (FBS), 1% sodium pyruvate, 1% HEPES, 1% L-glutamine, 1% penicillin streptomycin, and 42.4% of 4T1 conditioned medium was distributed equally into each well (~4.715 mL/well). Approximately 1.98 x 106 black-6 bone marrow derived stem cells extracted from tibias and femurs of a six-week old mouse were grown in this medium. .1ml (low concentration), 1.0ml (mid concentration), and 10.0ml (high concentration) of IL-6 neutralizing antibody were added to a Black-6 stem cell/4T1 conditioned medium complex. These cells then analyzed using a flow cytometer after an incubation period of six days. IL-6 concentration assay. The remaining growth medium used in the depletion assay was evenly distributed among seven plates (approximately 4.715 mL of culture medium per well). 1.98 x 106 black-6 stem cells were added to each well. Concentrations of 5ml (low concentration), 50ml (mid concentration), and 100ml (high concentration) were used. Cells were grown and analyzed using the same procedure as the IL-6 depletion assay. |